|
4/1/2023 0 Comments Spike protein vaccine 
Spike protein binding assays are one method in use by the Coronavirus Immunotherapy Consortium for assessing antibody treatments 6. In therapeutic monoclonal antibody development, standardization of binding assays is important for comparative data analysis. In addition to vaccine development, numerous efforts to produce large quantities of spike protein are underway in order to supply the high demand for therapeutic, diagnostic, and serosurveillance methods. Thus, recombinant proteins are a worthwhile complement to the novel technologies in parallel development 5. Recombinant spike protein constructs, both full length and soluble ectodomain, are the basis of candidates in late-stage clinical trials, including those sponsored by Novavax, Sanofi Pasteur, and GSK 3, 4, and have the benefit of robust commercial experience and previous licensure. 
We anticipate our results will provide a blueprint for worldwide biopharmaceutical production laboratories, as well as a starting point for process intensification.įollowing the emergence of the SARS-CoV-2 virus in late 2019, a platform approach to betacoronavirus spike protein stabilization in the pre-fusion conformation, along with early solved atomic-level structures of the stabilized spike, allowed for rapid selection of the SARS-CoV-2 spike protein as an antigen for vaccine development 1, 2. Our results demonstrate that commercially available chromatography resins are suitable for cGMP manufacturing of SARS-CoV-2 Spike protein constructs. The developed process was applied to purify HexaPro from a CHO-DG44 stable pool harvest and yielded the highest yet reported amount of pure S protein. The chromatographic unit operations along with industry-standard methodologies for viral clearances, low pH treatment and 20 nm filtration, were assessed for feasibility. A high-throughput resin screen on the Tecan Freedom EVO200 automated bioprocess workstation led to identification of ion exchange resins as viable purification steps. 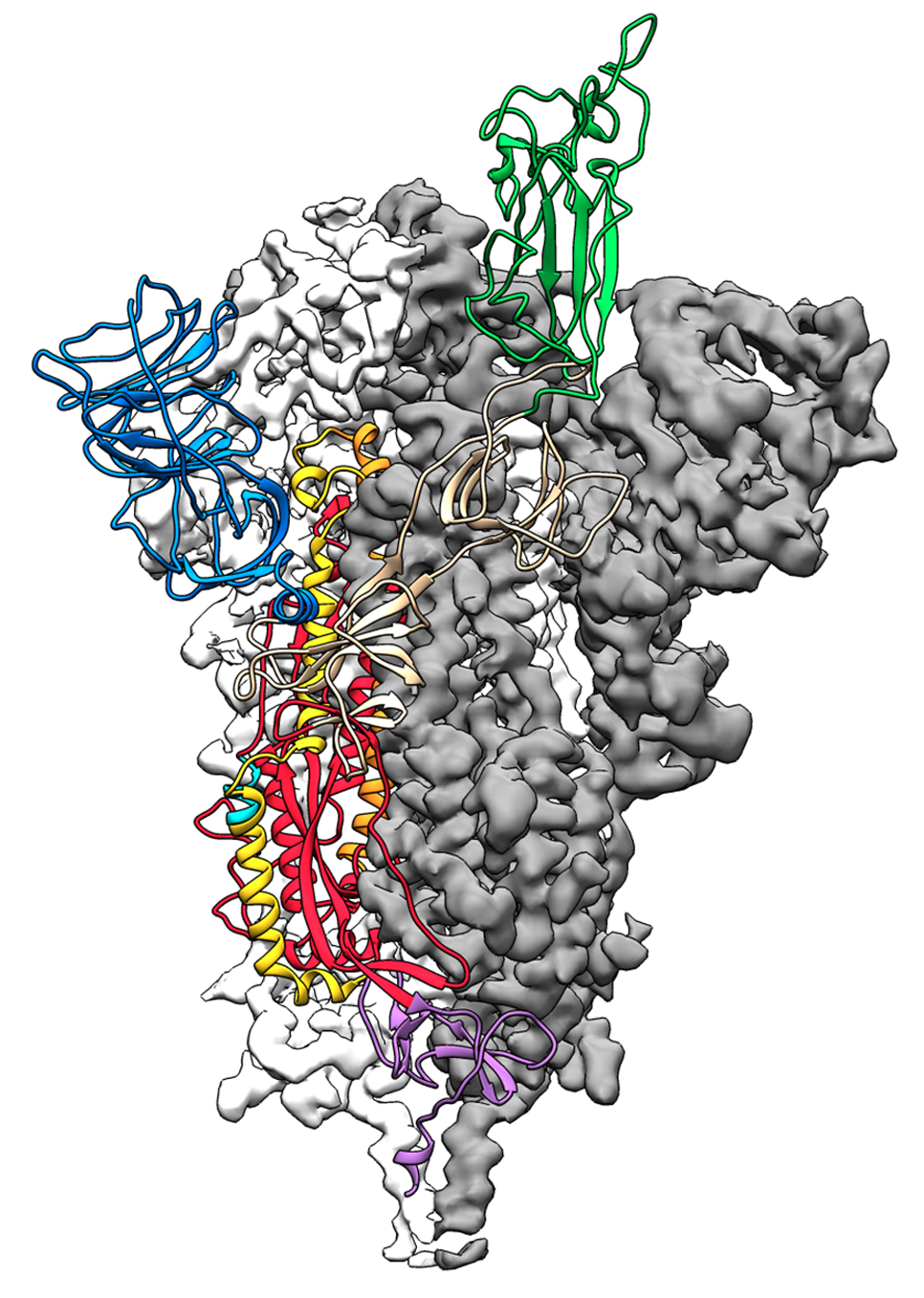
Herein we present the first known non-affinity purification method for two S constructs, S_dF_2P and HexaPro, expressed in the mammalian cell line, CHO-DG44. As of June 2021, S proteins have exclusively been purified using difficult-to-scale, low-yield methodologies such as affinity and size-exclusion chromatography. A high-yield, scalable, cGMP-compliant downstream process for the stabilized, soluble, native-like S protein ectodomain is necessary to meet the extensive material requirements for ongoing research and development. The spike (S) glycoprotein of the pandemic virus, SARS-CoV-2, is a critically important target of vaccine design and therapeutic development. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
